World Journal of Emergency Medicine ›› 2023, Vol. 14 ›› Issue (6): 421-427.doi: 10.5847/wjem.j.1920-8642.2023.093
• Review Article • Next Articles
Jingyi Wang1,2, Li Weng2, Jun Xu1( ), Bin Du2(
), Bin Du2( )
)
Received:2022-12-26
Accepted:2023-05-14
Online:2023-11-10
Published:2023-11-01
Contact:
Jun Xu, Email: Jingyi Wang, Li Weng, Jun Xu, Bin Du. Blood gas analysis as a surrogate for microhemodynamic monitoring in sepsis[J]. World Journal of Emergency Medicine, 2023, 14(6): 421-427.
Add to citation manager EndNote|Ris|BibTeX
URL: http://wjem.com.cn/EN/10.5847/wjem.j.1920-8642.2023.093
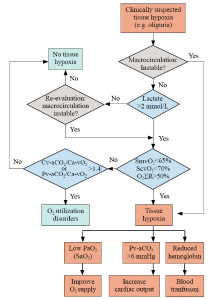
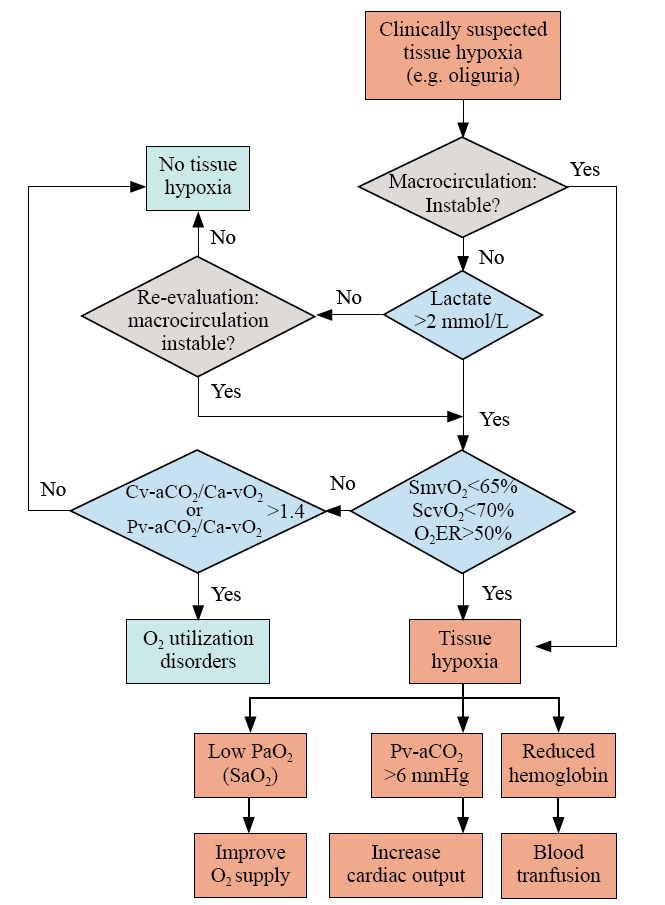
Figure 3.
Integrated interpretation of blood gas analysis in hemodynamic monitoring. O2ER: O2 extraction rate; SvO2: venous O2 saturation; Pv-aCO2: venous to arterial CO2 tension; Ca-vCO2: arterial to venous CO2 content; Ca-vO2: arterial to venous O2 content; SaO2: arterial O2 saturation; ScvO2: central venous O2 saturation; SmvO2: mixed venous O2 saturation.
| 1 |
Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021; 47(11): 1181-247.
doi: 10.1007/s00134-021-06506-y pmid: 34599691 |
| 2 |
Trzeciak S, McCoy JV, Phillip Dellinger R, Arnold RC, Rizzuto M, Abate NL, et al. Early increases in microcirculatory perfusion during protocol-directed resuscitation are associated with reduced multi-organ failure at 24 h in patients with sepsis. Intensive Care Med. 2008; 34(12): 2210-7.
doi: 10.1007/s00134-008-1193-6 |
| 3 |
De Backer D, Donadello K, Sakr Y, Ospina-Tascon G, Salgado D, Scolletta S, et al. Microcirculatory alterations in patients with severe sepsis: impact of time of assessment and relationship with outcome. Crit Care Med. 2013; 41(3): 791-9.
doi: 10.1097/CCM.0b013e3182742e8b pmid: 23318492 |
| 4 | De Backer D, Hollenberg S, Boerma C, Goedhart P, Büchele G, Ospina-Tascon G, et al. How to evaluate the microcirculation: report of a round table conference. Crit Care. 2007; 11(5): R101. |
| 5 |
De Backer D. Is microcirculatory assessment ready for regular use in clinical practice? Curr Opin Crit Care. 2019; 25(3): 280-4.
doi: 10.1097/MCC.0000000000000605 pmid: 31022089 |
| 6 | Cecconi M, De Backer D, Antonelli M, Beale R, Bakker J, Hofer C, et al. Consensus on circulatory shock and hemodynamic monitoring. Task force of the European Society of Intensive Care Medicine. Intensive Care Med. 2014; 40(12): 1795-815. |
| 7 |
Trzeciak S, Dellinger RP, Parrillo JE, Guglielmi M, Bajaj J, Abate NL, et al. Early microcirculatory perfusion derangements in patients with severe sepsis and septic shock: relationship to hemodynamics, oxygen transport, and survival. Ann Emerg Med. 2007; 49(1): 88-98, 98.e1-2.
pmid: 17095120 |
| 8 |
Sakr Y, Dubois MJ, De Backer D, Creteur J, Vincent JL. Persistent microcirculatory alterations are associated with organ failure and death in patients with septic shock. Crit Care Med. 2004; 32(9): 1825-31.
doi: 10.1097/01.ccm.0000138558.16257.3f pmid: 15343008 |
| 9 |
De Backer D, Creteur J, Preiser JC, Dubois MJ, Vincent JL. Microvascular blood flow is altered in patients with sepsis. Am J Respir Crit Care Med. 2002; 166(1): 98-104.
doi: 10.1164/rccm.200109-016OC |
| 10 |
Ince C. Hemodynamic coherence and the rationale for monitoring the microcirculation. Crit Care. 2015; 19(Suppl 3): S8.
doi: 10.1186/cc14726 |
| 11 |
Yajnik V, Maarouf R. Sepsis and the microcirculation: the impact on outcomes. Curr Opin Anaesthesiol. 2022; 35(2): 230-5.
doi: 10.1097/ACO.0000000000001098 pmid: 35081058 |
| 12 |
Arina P, Singer M. Pathophysiology of sepsis. Curr Opin Anaesthesiol. 2021; 34(2): 77-84.
doi: 10.1097/ACO.0000000000000963 pmid: 33652454 |
| 13 |
Mallat J, Lemyze M, Meddour M, Pepy F, Gasan G, Barrailler S, et al. Ratios of central venous-to-arterial carbon dioxide content or tension to arteriovenous oxygen content are better markers of global anaerobic metabolism than lactate in septic shock patients. Ann Intensive Care. 2016; 6(1): 10.
doi: 10.1186/s13613-016-0110-3 pmid: 26842697 |
| 14 | Lee CP, Bora V. Anesthesia monitoring of mixed venous saturation. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2022. |
| 15 |
Hartog C, Bloos F. Venous oxygen saturation. Best Pract Res Clin Anaesthesiol. 2014; 28(4): 419-28.
doi: 10.1016/j.bpa.2014.09.006 pmid: 25480771 |
| 16 |
van Beest PA, van Ingen J, Boerma EC, Holman ND, Groen H, Koopmans M, et al. No agreement of mixed venous and central venous saturation in sepsis, independent of sepsis origin. Crit Care. 2010; 14(6): R219.
doi: 10.1186/cc9348 |
| 17 |
Soussi MS, Jebali MA, Le Manach Y, Nasri M, Zouari B, Chenik S, et al. Central venous saturation is not an alternative to mixed venous saturation during cardiopulmonary bypass in coronary artery surgery patients. Perfusion. 2012; 27(4): 300-6.
doi: 10.1177/0267659112442902 pmid: 22499058 |
| 18 |
Lorentzen AG, Lindskov C, Sloth E, Jakobsen CJ. Central venous oxygen saturation cannot replace mixed venous saturation in patients undergoing cardiac surgery. J Cardiothorac Vasc Anesth. 2008; 22(6): 853-7.
doi: 10.1053/j.jvca.2008.04.004 |
| 19 |
Kopterides P, Bonovas S, Mavrou I, Kostadima E, Zakynthinos E, Armaganidis A. Venous oxygen saturation and lactate gradient from superior vena cava to pulmonary artery in patients with septic shock. Shock. 2009; 31(6): 561-7.
doi: 10.1097/SHK.0b013e31818bb8d8 pmid: 18838939 |
| 20 | Reinhart K, Kuhn HJ, Hartog C, Bredle DL. Continuous central venous and pulmonary artery oxygen saturation monitoring in the critically ill. Intensive Care Med. 2004; 30(8): 1572-8. |
| 21 |
He HW, Long Y, Zhou X, Wang XT, Zhang HM, Chai WZ, et al. Oxygen-flow-pressure targets for resuscitation in critical hemodynamic therapy. Shock. 2018; 49(1): 15-23.
doi: 10.1097/SHK.0000000000000929 pmid: 28650929 |
| 22 |
Squara P. Central venous oxygenation: when physiology explains apparent discrepancies. Crit Care. 2014; 18(6): 579.
doi: 10.1186/s13054-014-0579-9 |
| 23 |
Thooft A, Favory R, Salgado DR, Taccone FS, Donadello K, De Backer D, et al. Effects of changes in arterial pressure on organ perfusion during septic shock. Crit Care. 2011; 15(5): R222.
doi: 10.1186/cc10462 |
| 24 | Arango-Granados MC, Umaña M, Sánchez ÁI, García AF, Granados M, Ospina-Tascón GA. Impact of red blood cell transfusion on oxygen transport and metabolism in patients with sepsis and septic shock: a systematic review and meta-analysis. Rev Bras Ter Intensiva. 2021;33(1): 154-66. |
| 25 |
Textoris J, Fouché L, Wiramus S, Antonini F, Tho S, Martin C, et al. High central venous oxygen saturation in the latter stages of septic shock is associated with increased mortality. Crit Care. 2011; 15(4): R176.
doi: 10.1186/cc10325 |
| 26 |
Perz S, Uhlig T, Kohl M, Bredle DL, Reinhart K, Bauer M, et al. Low and “supranormal” central venous oxygen saturation and markers of tissue hypoxia in cardiac surgery patients: a prospective observational study. Intensive Care Med. 2011; 37(1): 52-9.
doi: 10.1007/s00134-010-1980-8 |
| 27 |
Wittayachamnankul B, Apaijai N, Sutham K, Chenthanakij B, Liwsrisakun C, Jaiwongkam T, et al. High central venous oxygen saturation is associated with mitochondrial dysfunction in septic shock: a prospective observational study. J Cell Mol Med. 2020; 24(11): 6485-94.
doi: 10.1111/jcmm.15299 pmid: 32352229 |
| 28 | Huber W, Zanner R, Schneider G, Schmid R, Lahmer T. Assessment of regional perfusion and organ function: less and non-invasive techniques. Front Med (Lausanne). 2019; 6: 50. |
| 29 |
Haase N, Perner A. Central venous oxygen saturation in septic shock—a marker of cardiac output, microvascular shunting and/or dysoxia? Crit Care. 2011; 15(4): 184.
doi: 10.1186/cc10314 |
| 30 |
He HW, Liu DW, Long Y, Wang XT. Mind the influence of arterial oxygen tension on central venous oxygen saturation. Crit Care. 2014; 18(5): 569.
doi: 10.1186/s13054-014-0569-y |
| 31 | Vincent JL, Theerawit P, Simion D. Basic principles of oxygen transport and calculations. Alternatives to Blood Transfusion in Transfusion Medicine. Oxford: Wiley-Blackwell, 2010: 203-9. |
| 32 |
Nasser B, Tageldein M, AlMesned A, Kabbani M. Effects of blood transfusion on oxygen extraction ratio and central venous saturation in children after cardiac surgery. Ann Saudi Med. 2017; 37(1): 31-7.
doi: 10.5144/0256-4947.2017.31 pmid: 28151454 |
| 33 |
An X, Zhang H, Sun YN, Ma XC. The microcirculatory failure could not weaken the increase of systematic oxygen extraction rate in septic shock: an observational study in canine models. Clin Hemorheol Microcirc. 2016; 63(3): 267-79.
doi: 10.3233/CH-152022 pmid: 26639767 |
| 34 | Ospina-Tascón GA, García Marin AF, Echeverri GJ, Bermudez WF, Madriñán-Navia H, Valencia JD, et al. Effects of dobutamine on intestinal microvascular blood flow heterogeneity and O2 extraction during septic shock. J Appl Physiol (1985). 2017;122(6): 1406-17. |
| 35 |
Levy B. Lactate and shock state: the metabolic view. Curr Opin Crit Care. 2006; 12(4): 315-21.
doi: 10.1097/01.ccx.0000235208.77450.15 pmid: 16810041 |
| 36 |
Kraut JA, Madias NE. Lactic acidosis. N Engl J Med. 2014; 371(24): 2309-19.
doi: 10.1056/NEJMra1309483 |
| 37 |
Levy B, Desebbe O, Montemont C, Gibot S. Increased aerobic glycolysis through beta2 stimulation is a common mechanism involved in lactate formation during shock states. Shock. 2008; 30(4): 417-21.
doi: 10.1097/SHK.0b013e318167378f pmid: 18323749 |
| 38 | Taylor DJ, Faragher EB, Evanson JM. Inflammatory cytokines stimulate glucose uptake and glycolysis but reduce glucose oxidation in human dermal fibroblasts in vitro. Circ Shock. 1992; 37(2): 105-10. |
| 39 |
Ospina-Tascon G, Neves AP, Occhipinti G, Donadello K, Büchele G, Simion D, et al. Effects of fluids on microvascular perfusion in patients with severe sepsis. Intensive Care Med. 2010; 36(6): 949-55.
doi: 10.1007/s00134-010-1843-3 pmid: 20221744 |
| 40 |
Filbin MR, Hou PC, Massey M, Barche A, Kao E, Bracey A, et al. The microcirculation is preserved in emergency department low-acuity sepsis patients without hypotension. Acad Emerg Med. 2014; 21(2): 154-62.
doi: 10.1111/acem.12314 pmid: 24673671 |
| 41 |
Puskarich MA, Shapiro NI, Massey MJ, Kline JA, Jones AE. Lactate clearance in septic shock is not a surrogate for improved microcirculatory flow. Acad Emerg Med. 2016; 23(6): 690-3.
doi: 10.1111/acem.12928 pmid: 26825368 |
| 42 | Patel H, Kerndt CC, Bhardwaj A. Physiology, respiratory quotient. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2022. |
| 43 | Randall HM Jr, Cohen JJ. Anaerobic CO2 production by dog kidney in vitro. Am J Physiol Leg Content. 1966; 211(2): 493-505. |
| 44 |
da Rocha EE, Alves VG, da Fonseca RB. Indirect calorimetry: methodology, instruments and clinical application. Curr Opin Clin Nutr Metab Care. 2006; 9(3): 247-56.
doi: 10.1097/01.mco.0000222107.15548.f5 |
| 45 |
De Waele E, Honore PM, Spapen HD. New generation indirect calorimeters for measuring energy expenditure in the critically ill: a rampant or reticent revolution? Crit Care. 2016; 20(1): 138.
doi: 10.1186/s13054-016-1315-4 |
| 46 |
Ltaief Z, Schneider AG, Liaudet L. Pathophysiology and clinical implications of the veno-arterial PCO2 gap. Crit Care. 2021; 25(1): 318.
doi: 10.1186/s13054-021-03671-w |
| 47 |
Mekontso-Dessap A, Castelain V, Anguel N, Bahloul M, Schauvliege F, Richard C, et al. Combination of venoarterial PCO2 difference with arteriovenous O2 content difference to detect anaerobic metabolism in patients. Intensive Care Med. 2002; 28(3): 272-7.
doi: 10.1007/s00134-002-1215-8 |
| 48 |
Mesquida J, Saludes P, Gruartmoner G, Espinal C, Torrents E, Baigorri F, et al. Central venous-to-arterial carbon dioxide difference combined with arterial-to-venous oxygen content difference is associated with lactate evolution in the hemodynamic resuscitation process in early septic shock. Crit Care. 2015; 19(1): 126.
doi: 10.1186/s13054-015-0858-0 |
| 49 | He H, Long Y, Liu D, Wang X, Tang B. The prognostic value of central venous-to-arterial CO2 difference/arterial-central venous O2 difference ratio in septic shock patients with central venous O2 saturation ≥80. Shock. 2017;48(5): 551-7. |
| 50 | Ospina-Tascón GA, Umaña M, Bermúdez W, Bautista-Rincón DF, Hernandez G, Bruhn A, et al. Combination of arterial lactate levels and venous-arterial CO2 to arterial-venous O2 content difference ratio as markers of resuscitation in patients with septic shock. Intensive Care Med. 2015;41(5): 796-805. |
| 51 | Ospina-Tascón GA, Umaña M, Bermúdez WF, Bautista-Rincón DF, Valencia JD, Madriñán HJ, et al. Can venous-to-arterial carbon dioxide differences reflect microcirculatory alterations in patients with septic shock? Intensive Care Med. 2016;42(2): 211-21. |
| 52 | He HW, Liu DW, Long Y, Wang XT, Yu C, Yao B, et al. Using peripheral perfusion index and venous-to-arterial CO(2) difference/arterial-central venous O(2) difference ratio to assess lactate clearance in septic patients after resuscitation. Zhonghua Nei Ke Za Zhi. 2018;57(12): 917-21. [Article in Chinese] |
| 53 | Troskot R, Šimurina T, Žižak M, Majstorović K, Marinac I, Mrakovčić-Šutić I. Prognostic value of venoarterial carbon dioxide gradient in patients with severe sepsis and septic shock. Croat Med J. 2010;51(6): 501-8. |
| 54 |
Mallat J, Pepy F, Lemyze M, Gasan G, Vangrunderbeeck N, Tronchon L, et al. Central venous-to-arterial carbon dioxide partial pressure difference in early resuscitation from septic shock: a prospective observational study. Eur J Anaesthesiol. 2014; 31(7): 371-80.
doi: 10.1097/EJA.0000000000000064 pmid: 24625464 |
| 55 |
Guo ZQ, Yin M, Kong JC, Wang B, Dai KP, Zuo T, et al. Relationship analysis of central venous-to-arterial carbon dioxide difference and cardiac index for septic shock. Sci Rep. 2019; 9(1): 8822.
doi: 10.1038/s41598-019-45252-6 pmid: 31217467 |
| 56 |
Dres M, Monnet X, Teboul JL. Hemodynamic management of cardiovascular failure by using PCO(2) venous-arterial difference. J Clin Monit Comput. 2012; 26(5): 367-74.
doi: 10.1007/s10877-012-9381-x pmid: 22828858 |
| 57 |
Mallat J, Lemyze M, Tronchon L, Vallet B, Thevenin D. Use of venous-to-arterial carbon dioxide tension difference to guide resuscitation therapy in septic shock. World J Crit Care Med. 2016; 5(1): 47-56.
doi: 10.5492/wjccm.v5.i1.47 pmid: 26855893 |
| 58 |
Durkin R, Gergits MA, Reed JF 3rd, Fitzgibbons J. The relationship between the arteriovenous carbon dioxide gradient and cardiac index. J Crit Care. 1993; 8(4): 217-21.
pmid: 8305959 |
| 59 |
Habicher M, von Heymann C, Spies CD, Wernecke KD, Sander M. Central venous-arterial pCO2 difference identifies microcirculatory hypoperfusion in cardiac surgical patients with normal central venous oxygen saturation: a retrospective analysis. J Cardiothorac Vasc Anesth. 2015; 29(3): 646-55.
doi: 10.1053/j.jvca.2014.09.006 pmid: 25575410 |
| [1] | Saifeng Chen, Xuewei Hao, Guo Chen, Guorong Liu, Xiaoyan Yuan, Peiling Shen, Dongfeng Guo. Effects of mesencephalic astrocyte-derived neurotrophic factor on sepsis-associated acute kidney injury [J]. World Journal of Emergency Medicine, 2023, 14(5): 386-392. |
| [2] | Mubing Qin, Yanxia Gao, Shigong Guo, Xin Lu, Qian Zhao, Zengzheng Ge, Huadong Zhu, Yi Li. Establishment and evaluation of animal models of sepsis-associated encephalopathy [J]. World Journal of Emergency Medicine, 2023, 14(5): 349-353. |
| [3] | Meng-meng An, Chen-xi Liu, Ping Gong. Effects of continuous renal replacement therapy on inflammation-related anemia, iron metabolism and prognosis in sepsis patients with acute kidney injury [J]. World Journal of Emergency Medicine, 2023, 14(3): 186-192. |
| [4] | Jue-xian Wei, Hui-lin Jiang, Xiao-hui Chen. Endothelial cell metabolism in sepsis [J]. World Journal of Emergency Medicine, 2023, 14(1): 10-16. |
| [5] | Hui Liu, Jie Hu, Jian-guo Xiao, Hong-jun Kang, Fei-hu Zhou. The procalcitonin-to-cortisol ratio is a potential prognostic predictor in sepsis with abdominal source: a retrospective observational study [J]. World Journal of Emergency Medicine, 2022, 13(6): 441-447. |
| [6] | Ralph Bou Chebl, Nadim Kattouf, Mohamad Assaf, Saadeddine Haidar, Gilbert Abou Dagher, Sarah Abdul Nabi, Rana Bachir, Mazen El Sayed. Comparing the demographic data and outcomes of septic shock patients presenting to teaching or non-teaching metropolitan hospitals in the United States [J]. World Journal of Emergency Medicine, 2022, 13(6): 433-440. |
| [7] | A-ling Tang, Mei-jia Shen, Guo-qiang Zhang. Intestinal microcirculation dysfunction in sepsis: pathophysiology, clinical monitoring, and therapeutic interventions [J]. World Journal of Emergency Medicine, 2022, 13(5): 343-348. |
| [8] | Shi-yuan Yu, Zeng-zheng Ge, Jun Xiang, Yan-xia Gao, Xin Lu, Joseph Harold Walline, Mu-bing Qin, Hua-dong Zhu, Yi Li. Is rosuvastatin protective against sepsis-associated encephalopathy? A secondary analysis of the SAILS trial [J]. World Journal of Emergency Medicine, 2022, 13(5): 367-372. |
| [9] | Xiao-kang Dai, Zhen-xing Ding, Yuan-yuan Tan, Hua-rui Bao, Dong-yao Wang, Hong Zhang. Neutrophils inhibit CD8+ T cells immune response by arginase-1 signaling in patients with sepsis [J]. World Journal of Emergency Medicine, 2022, 13(4): 266-273. |
| [10] | Xuan Fu, Xue Lin, Samuel Seery, Li-na Zhao, Hua-dong Zhu, Jun Xu, Xue-zhong Yu. Speckle-tracking echocardiography for detecting myocardial dysfunction in sepsis and septic shock patients: A single emergency department study [J]. World Journal of Emergency Medicine, 2022, 13(3): 175-181. |
| [11] | Mei-jia Shen, Li-chao Sun, Xiao-yu Liu, Meng-chen Xiong, Shan Li, A-ling Tang, Guo-qiang Zhang. Trichostatin A improves the inflammatory response and liver injury in septic mice through the FoxO3a/autophagy signaling pathway [J]. World Journal of Emergency Medicine, 2022, 13(3): 182-188. |
| [12] | Hai Hu, Jing-yuan Jiang, Ni Yao. Comparison of different versions of the quick sequential organ failure assessment for predicting in-hospital mortality of sepsis patients: A retrospective observational study [J]. World Journal of Emergency Medicine, 2022, 13(2): 114-119. |
| [13] | Li-wei Duan, Jin-long Qu, Jian Wan, Yong-hua Xu, Yi Shan, Li-xue Wu, Jin-hao Zheng, Wei-wei Jiang, Qi-tong Chen, Yan Zhu, Jian Zhou, Wen-bo Yu, Lei Pei, Xi Song, Wen-fang Li, Zhao-fen Lin. Effects of viral infection and microbial diversity on patients with sepsis: A retrospective study based on metagenomic next-generation sequencing [J]. World Journal of Emergency Medicine, 2021, 12(1): 29-35. |
| [14] | Hai-jiang Zhou, Tian-fei Lan, Shu-bin Guo. Outcome prediction value of National Early Warning Score in septic patients with community-acquired pneumonia in emergency department: A single-center retrospective cohort study [J]. World Journal of Emergency Medicine, 2020, 11(4): 206-215. |
| [15] | Yu-ming Wang, Yan-jun Zheng, Ying Chen, Yun-chuan Huang, Wei-wei Chen, Ran Ji, Li-li Xu, Zhi-tao Yang, Hui-qiu Sheng, Hong-ping Qu, En-qiang Mao, Er-zhen Chen. Effects of fluid balance on prognosis of acute respiratory distress syndrome patients secondary to sepsis [J]. World Journal of Emergency Medicine, 2020, 11(4): 216-222. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
