World Journal of Emergency Medicine ›› 2020, Vol. 11 ›› Issue (2): 79-86.doi: 10.5847/wjem.j.1920-8642.2020.02.003
Special Issue: Sepsis
• Original Articles • Previous Articles Next Articles
Wen-peng Yin, Jia-bao Li, Xiao-fang Zheng, Le An, Huan Shao, Chun-sheng Li( )
)
Received:2019-08-12
Accepted:2019-12-20
Online:2020-04-01
Published:2020-04-01
Contact:
Chun-sheng Li
E-mail:lcscyyy@163.com
Wen-peng Yin, Jia-bao Li, Xiao-fang Zheng, Le An, Huan Shao, Chun-sheng Li. Effect of neutrophil CD64 for diagnosing sepsis in emergency department[J]. World Journal of Emergency Medicine, 2020, 11(2): 79-86.
Add to citation manager EndNote|Ris|BibTeX
URL: http://wjem.com.cn//EN/10.5847/wjem.j.1920-8642.2020.02.003
Table 1
Baseline characteristics of patients based on severity of sepsis
| Variables | Control (n=20) | Sepsis (n=119) | Septic shock (n=32) | P value |
|---|---|---|---|---|
| Age, years | 62.5±6.3 | 65.2±15.9 | 66.3±15.2 | 0.332a |
| Male, n (%) | 12 (60.0) | 77 (64.7) | 26 (81.3) | 0.074b |
| Mortality, n (%) | 0 | 15 (12.6) | 15 (46.9) | 0.000 |
| Infection site, n (%) | ||||
| Respiratory | 0 | 86 (72.3) | 26 (81.3) | 0.303 |
| hepatobilinary | 0 | 6 (5.0) | 2 (6.3) | 0.677 |
| Urinary | 0 | 11 (9.2) | 2 (6.3) | 0.736 |
| Gastrointestinal | 0 | 14 (11.8) | 2 (6.3) | 0.525 |
| Cutaneous | 0 | 2 (1.7) | 0 | 1.000 |
| Comorbidity, n (%) | ||||
| COPD | 0 | 36 (30.3) | 15 (46.9) | 0.078 |
| Diabetes | 0 | 28 (23.5) | 19 (59.4) | 0.000 |
| CDVD | 0 | 32 (26.9) | 12 (37.5) | 0.241 |
| CBVD | 0 | 26 (21.8) | 8 (25.0) | 0.705 |
| CRD | 0 | 15 (12.6) | 6 (18.8) | 0.393 |
| HBD | 0 | 13 (10.9) | 3 (9.4) | 1.000 |
| Healthy | 0 | 11 (9.2) | 7 (21.9) | 0.065 |
| Positive culture, n | ||||
| Klebsiella pneumoniae | 0 | 4 | 3 | |
| Streptococcus pneumoniae | 0 | 2 | 2 | |
| Staphylococcus aureus | 0 | 3 | 3 | |
| Pseudomonas aeruginosa | 0 | 2 | 3 | |
| Escherichia coli | 0 | 1 | 4 | |
| Enterobactor cloacae | 0 | 1 | 0 | |
| SOFA | 0 | 3 (3, 4) | 6 (3, 8) | <0.001 |
| CD64 (MFI) | 2.2 (2.0, 2.5) | 4.1 (3.1, 6.6) | 9 (6.4, 14.8) | <0.001 |
| PCT (ng/mL) | 0.04 (0.02, 0.06) | 1.8 (0.4, 7.0) | 17.1 (6.7, 45.0) | <0.001 |
| CRP (mg/L) | 4.6 (3.9, 6.2) | 13.6 (12.6, 14.6) | 14.6 (12.4, 15.7) | 0.086 |
| WBC (×109/L) | 5.7 (5.0, 6.7) | 12.0 (9.1, 15.3) | 12.9 (7.9, 19.1) | 0.503 |
Table 2
Analysis of ROC curves in diagnosing positive infection culture in patients with sepsis
| Variables | AUC (95% CI) | P value | Cut-off value | Sensitivity | Specificity | PPV | NPV |
|---|---|---|---|---|---|---|---|
| SOFA | 0.701 (0.579-0.824) | 0.001 | 5.5 | 0.571 | 0.829 | 0.431 | 0.895 |
| CD64 | 0.879 (0.795-0.962) | <0.001 | 8 | 0.750 | 0.894 | 0.616 | 0.940 |
| PCT | 0.868 (0.798-0.937) | <0.001 | 4.85 | 0.929 | 0.699 | 0.412 | 0.977 |
| CRP | 0.609 (0.491-0.727) | 0.071 | 14.69 | 0.464 | 0.772 | 0.316 | 0.864 |
| WBC | 0.525 (0.399-0.651) | 0.681 | 15.14 | 0.429 | 0.748 | 0.279 | 0.852 |
| CD64+SOFA | 0.888 (0.814-0.962) | <0.001 | 0.195 | 0.821 | 0.829 | 0.521 | 0.953 |
| PCT+SOFA | 0.848 (0.752-0.945) | <0.001 | 0.164 | 0.821 | 0.813 | 0.499 | 0.952 |
| CRP+SOFA | 0.716 (0.596-0.835) | <0.001 | 0.227 | 0.571 | 0.829 | 0.431 | 0.895 |
| WBC+SOFA | 0.700 (0.578-0.822) | 0.001 | 0.247 | 0.571 | 0.837 | 0.443 | 0.896 |
Table 3
Baseline characteristics of patients based on outcome
| Variables | Survival (n=121) | Death (n=30) | P value |
|---|---|---|---|
| Age, years | 64.3 (55.7, 79.2) | 72.5 (62.0, 80.3) | 0.052 |
| Male, n (%) | 84 (69.4) | 19 (63.3) | 0.521 |
| Infection site, n (%) | |||
| Respiratory | 88 (72.7) | 24 (80.0) | 0.415 |
| hepatobilinary | 6 (5.0) | 2 (6.7) | 0.659 |
| Urinary | 11 (9.1) | 2 (6.7) | 1.000 |
| Gastrointestinal | 14 (11.6) | 2 (6.7) | 0.740 |
| Cutaneous | 2 (1.7) | 0 | 1.000 |
| Comorbidity, n (%) | |||
| COPD | 38 (31.4) | 13 (43.3) | 0.216 |
| Diabetes | 35 (28.9) | 12 (40.0) | 0.241 |
| CDVD | 36 (29.8) | 8 (26.7) | 0.739 |
| CBVD | 32 (26.4) | 2 (6.7) | 0.020 |
| CRD | 13 (10.7) | 8 (26.7) | 0.037 |
| HBD | 11 (9.1) | 2 (6.7) | 1.000 |
| Healthy | 13 (10.7) | 5 (16.7) | 0.357 |
| Positive culture, n | |||
| Klebsiella pneumoniae | 3 | 4 | |
| Streptococcus pneumoniae | 3 | 1 | |
| Staphylococcus aureus | 4 | 2 | |
| Pseudomonas aeruginosa | 3 | 2 | |
| Escherichia coli | 2 | 3 | |
| Enterobactor cloacae | 1 | 0 | |
| SOFA | 3 (3, 4) | 7.5 (5.8, 9) | <0.001 |
| CD64 (MFI) | 4.1 (3.1, 6.6) | 8.9 (2.9, 10.8) | <0.001 |
| PCT (ng/mL) | 1.8 (0.4, 8.8) | 9.2 (3.1, 20.7) | 0.001 |
| CRP (mg/L) | 13.6 (12.4, 14.7) | 14.5 (13.1, 15.7) | 0.038 |
| WBC (×109/L) | 12.4 (9, 15.4) | 11.1 (7.4, 20.3) | 0.618 |

Figure 2.
The ROC curves of nCD64, PCT, CRP, WBC and SOFA score for prognosis. The AUC of SOFA was the highest (0.889), followed by nCD64 (0.850), PCT (0.700), CRP (0.622) and WBC (0.529). There were significant differences between SOFA and CRP or PCT (P<0.001), same with the nCD64 (P<0.001), but there were no significant differences between SOFA and nCD64 (P=0.358), CRP and PCT (P=0.2637).
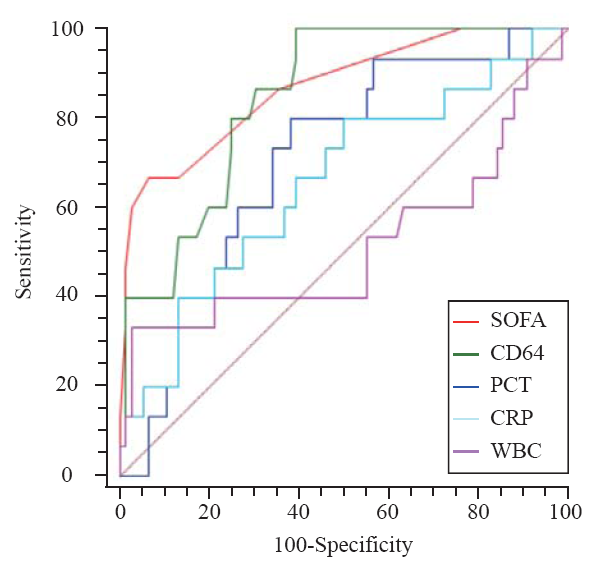
Table 4
Analysis of ROC curves in predicting 28-day mortality in patients with sepsis
| Variables | AUC (95% CI) | P value | Cut-off value | Sensitivity | Specificity | PPV | NPV |
|---|---|---|---|---|---|---|---|
| SOFA | 0.889 (0.821-0.958) | 0.000 | 5.500 | 0.767 | 0.884 | 0.621 | 0.939 |
| CD64 | 0.850 (0.786-0.914) | 0.000 | 5.450 | 0.933 | 0.653 | 0.400 | 0.975 |
| PCT | 0.700 (0.606-0.759) | 0.001 | 6.470 | 0.700 | 0.686 | 0.356 | 0.902 |
| CRP | 0.622 (0.505-0.740) | 0.038 | 14.050 | 0.633 | 0.612 | 0.288 | 0.871 |
| WBC | 0.529 (0.398-0.661) | 0.618 | 21.645 | 0.233 | 0.950 | 0.536 | 0.833 |
| CD64+SOFA | 0.916 (0.857-0.976) | 0.000 | 0.369 | 0.800 | 0.950 | 0.799 | 0.950 |
| PCT+SOFA | 0.882 (0.812-0.952) | 0.000 | 0.288 | 0.767 | 0.901 | 0.658 | 0.940 |
| CRP+SOFA | 0.895 (0.829-0.961) | 0.000 | 0.262 | 0.767 | 0.884 | 0.621 | 0.939 |
| WBC+SOFA | 0.890 (0.817-0.963) | 0.000 | 0.218 | 0.800 | 0.868 | 0.600 | 0.946 |
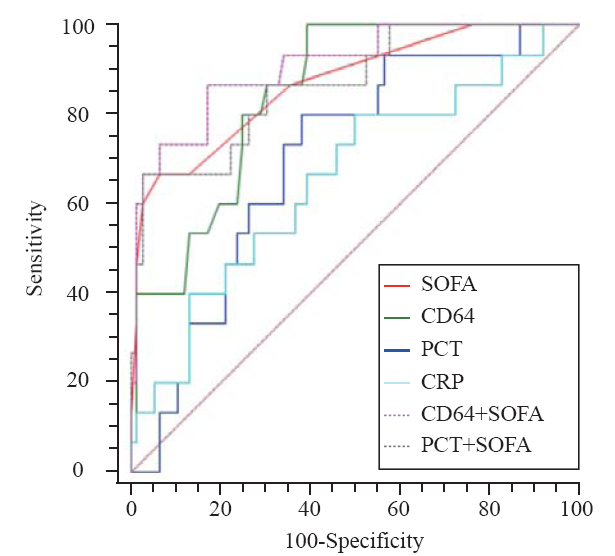
Figure 3.
The ROC curves of combination. The combination of nCD64 and SOFA achieved an AUC of 0.916, followed by the combination of PCT and SOFA (0.882). A significant difference of AUC was found between PCT+SOFA and PCT (P=0.0015), nCD64+SOFA and nCD64 (P=0.0160). There was no significant difference between nCD64+SOFA and PCT+SOFA (P=0.2028), nCD64+SOFA and SOFA (P=0.2366), PCT+SOFA and SOFA (P=0.5201), PCT+SOFA and CD64 (P=0.4804).
| 1 |
Meyer N, Harhay MO, Small DS, Prescott HC, Bowles KH, Gaieski DF, et al. Temporal trends in incidence, sepsis-related mortality, and hospital-based acute care after sepsis. Crit Care Med. 2018; 46(3):354-60.
doi: 10.1097/CCM.0000000000002872 pmid: 29474320 |
| 2 |
Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, Sharma S, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006; 34(6):1589-96.
doi: 10.1097/01.CCM.0000217961.75225.E9 pmid: 16625125 |
| 3 |
Kumar A, Ellis P, Arabi Y, Roberts D, Light B, Parrillo JE, et al. Initiation of inappropriate antimicrobial therapy results in a fivefold reduction of survival in human septic shock. Chest. 2009; 136(5):1237-48.
pmid: 19696123 |
| 4 |
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016; 315(8):801-10.
doi: 10.1001/jama.2016.0287 pmid: 26903338 |
| 5 |
Peters RP, van Agtmael MA, Danner SA, Savelkoul PH, Vandenbroucke-Grauls CM. New developments in the diagnosis of blood stream infections. Lancet Infect Dis. 2004; 4(12):751-60.
pmid: 15567125 |
| 6 |
van Engelen TSR, Wiersinga WJ, Scicluna BP, van der Poll T. Biomarkers in sepsis. Crit Care Clin. 2018; 34(1):139-52.
doi: 10.1016/j.ccc.2017.08.010 pmid: 29149935 |
| 7 |
Repp R, Valerius T, Sendler A, Gramatzki M, Iro H, Kalden JR, et al. Neutrophils express the high affinity receptor for IgG (Fc gamma RI, CD64) after in vivo application of recombinant human granulocyte colony-stimulating factor. Blood. 1991; 78(4):885-9.
pmid: 1714327 |
| 8 |
Davis BH, Olsen SH, Ahmad E, Bigelow NC. Neutrophil CD64 is an improved indicator of infection or sepsis in emergency department patients. Arch Pathol Lab Med. 2006; 130(5):654-61.
doi: 10.1043/1543-2165(2006)130[654:NCIAII]2.0.CO;2 pmid: 16683883 |
| 9 |
Pourmand A, Pyle M, Yamane D, Sumon K, Frasure SE. The utility of point-of-care ultrasound in the assessment of volume status in acute and critically ill patients. World J Emerg Med. 2019; 10(4):232-8.
pmid: 31534598 |
| 10 |
Bhandari V, Wang C, Rinder C, Rinder H. Hematologic profile of sepsis in neonates: neutrophil CD64 as a diagnostic marker. Pediatrics. 2008; 121(1):129-34.
pmid: 18166566 |
| 11 |
Wang X, Li ZY, Zeng L, Zhang AQ, Pan W, Gu W, et al. Neutrophil CD64 expression as a diagnostic marker for sepsis in adult patients: a meta-analysis. Crit Care. 2015; 19:245.
pmid: 26059345 |
| 12 |
Gros A, Roussel M, Sauvadet E, Gacouin A, Marque S, Chimot L, et al. The sensitivity of neutrophil CD64 expression as a biomarker of bacterial infection is low in critically ill patients. Intensive Care Med. 2012; 38(3):445-52.
doi: 10.1007/s00134-012-2483-6 pmid: 22310872 |
| 13 |
Yeh CF, Wu CC, Liu SH, Chen KF. Comparison of the accuracy of neutrophil CD64, procalcitonin, and C-reactive protein for sepsis identification: a systematic review and meta-analysis. Ann Intensive Care. 2019; 9(1):5.
doi: 10.1186/s13613-018-0479-2 |
| 14 |
Chambers KA, Park AY, Banuelos RC, Darger BF, Akkanti BH, Macaluso A, et al. Outcomes of severe sepsis and septic shock patients after stratification by initial lactate value. World J Emerg Med. 2018; 9(2):113-7.
pmid: 29576823 |
| 15 | Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med. 2017; 43(3):304-77. |
| 16 |
Raith EP, Udy AA, Bailey M, McGloughlin S, MacIsaac C, Bellomo R, et al. Prognostic accuracy of the SOFA Score, SIRS Criteria, and qSOFA Score for in-hospital mortality among adults with suspected infection admitted to the intensive care unit. JAMA. 2017; 317(3):290-300.
pmid: 28114553 |
| 17 |
Au-Yong A. Towards evidence-based emergency medicine: best BETs from the Manchester Royal Infirmary. BET 2: C-reactive protein in the diagnosis of bacteraemia. Emerg Med J. 2012; 29(5):423-4.
pmid: 22523145 |
| 18 |
Luzzani A, Polati E, Dorizzi R, Rungatscher A, Pavan R, Merlini A. Comparison of procalcitonin and C-reactive protein as markers of sepsis. Crit Care Med. 2003; 31(6):1737-41.
doi: 10.1097/01.CCM.0000063440.19188.ED pmid: 12794413 |
| 19 |
Koeze J, Hendrix MG, van den Bergh FA, Brouwer RM, Zijlstra JG. In critically ill patients the procalcitonin level can be misleading. Crit Care. 2011; 15(2):422.
doi: 10.1186/cc10132 pmid: 21542896 |
| 20 |
Porges AJ, Redecha PB, Doebele R, Pan LC, Salmon JE, Kimberly RP. Novel Fc gamma receptor I family gene products in human mononuclear cells. J Clin Invest. 1992; 90(5):2102-9.
doi: 10.1172/JCI116094 pmid: 1430234 |
| 21 |
Allen JM, Seed B. Isolation and expression of functional high-affinity Fc receptor complementary DNAs. Science. 1989; 243(4889):378-81.
pmid: 2911749 |
| 22 |
Yang AP, Liu J, Yue LH, Wang HQ, Yang WJ, Yang GH. Neutrophil CD64 combined with PCT, CRP and WBC improves the sensitivity for the early diagnosis of neonatal sepsis. Clin Chem Lab Med. 2016; 54(2):345-51.
pmid: 26351925 |
| 23 |
Kipfmueller F, Schneider J, Prusseit J, Dimitriou I, Zur B, Franz AR, et al. Role of neutrophil CD64 index as a screening marker for late-onset sepsis in very low birth weight infants. PLoS One. 2015; 10(4):e0124634.
doi: 10.1371/journal.pone.0124634 |
| 24 |
Groselj-Grenc M, Ihan A, Pavcnik-Arnol M, Kopitar AN, Gmeiner-Stopar T, Derganc M. Neutrophil and monocyte CD64 indexes, lipopolysaccharide-binding protein, procalcitonin and C-reactive protein in sepsis of critically ill neonates and children. Intensive Care Med. 2009; 35(11):1950-8.
doi: 10.1007/s00134-009-1637-7 pmid: 19756501 |
| 25 |
Rogina P, Stubljar D, Lejko-Zupanc T, Osredkar J, Skvarc M. Expression of CD64 on neutrophils (CD64 index): diagnostic accuracy of CD64 index to predict sepsis in critically ill patients. Clin Chem Lab Med. 2015; 53(4):e89-91.
pmid: 25324452 |
| 26 |
Cardelli P, Ferraironi M, Amodeo R, Tabacco F, De Blasi RA, Nicoletti M, et al. Evaluation of neutrophil CD64 expression and procalcitonin as useful markers in early diagnosis of sepsis. Int J Immunopathol Pharmacol. 2008; 21(1):43-9.
doi: 10.1177/039463200802100106 pmid: 18336730 |
| 27 |
Ma Y, Yu XY, Wang Y. Dose-related effects of dexmedetomidine on immunomodulation and mortality to septic shock in rats. World J Emerg Med. 2018; 9(1):56-63.
doi: 10.5847/wjem.j.1920-8642.2018.01.009 pmid: 29290897 |
| 28 |
de Jong E, de Lange DW, Beishuizen A, van de Ven PM, Girbes AR, Huisman A. Neutrophil CD64 expression as a longitudinal biomarker for severe disease and acute infection in critically ill patients. Int J Lab Hematol. 2016; 38(5):576-84.
doi: 10.1111/ijlh.12545 pmid: 27565453 |
| 29 |
Hoffmann JJ. Neutrophil CD64: a diagnostic marker for infection and sepsis. Clin Chem Lab Med. 2009; 47(8):903-16.
pmid: 19642859 |
| 30 |
Bae MH, Park SH, Park CJ, Cho EJ, Lee BR, Kim YJ, et al. Flow cytometric measurement of respiratory burst activity and surface expression of neutrophils for septic patient prognosis. Cytometry B Clin Cytom. 2016; 90(4):368-75.
doi: 10.1002/cyto.b.21274 pmid: 26202936 |
| 31 |
Danikas DD, Karakantza M, Theodorou GL, Sakellaropoulos GC, Gogos CA. Prognostic value of phagocytic activity of neutrophils and monocytes in sepsis. Correlation to CD64 and CD14 antigen expression. Clin Exp Immunol. 2008; 154(1):87-97.
doi: 10.1111/j.1365-2249.2008.03737.x pmid: 18727624 |
| 32 |
Djordjevic D, Pejovic J, Surbatovic M, Jevdjic J, Radakovic S, Veljovic M, et al. Prognostic value and daily trend of interleukin-6, neutrophil CD64 expression, c-reactive protein and lipopolysaccharide-binding protein in critically ill patients: reliable predictors of outcome or not? J Med Biochem. 2015; 34(4):431-9.
pmid: 28356852 |
| 33 |
Hsu KH, Chan MC, Wang JM, Lin LY, Wu CL. Comparison of Fcgamma receptor expression on neutrophils with procalcitonin for the diagnosis of sepsis in critically ill patients. Respirology. 2011; 16(1):152-60.
pmid: 20946336 |
| 34 |
Liu Y, Hou JH, Li Q, Chen KJ, Wang SN, Wang JM. Biomarkers for diagnosis of sepsis in patients with systemic inflammatory response syndrome: a systematic review and meta-analysis. Springerplus. 2016; 5(1):2091.
doi: 10.1186/s40064-016-3591-5 pmid: 28028489 |
| 35 |
Bauer PR, Kashyap R, League SC, Park JG, Block DR, Baumann NA, et al. Diagnostic accuracy and clinical relevance of an inflammatory biomarker panel for sepsis in adult critically ill patients. Diagn Microbiol Infect Dis. 2016; 84(2):175-80.
pmid: 26586579 |
| 36 |
Zhou H, Guo S, Lan T, Ma S, Zhang F, Zhao Z. Risk stratification and prediction value of procalcitonin and clinical severity scores for community-acquired pneumonia in ED. Am J Emerg Med. 2018; 36(12):2155-60.
pmid: 29691103 |
| 37 |
Dimoula A, Pradier O, Kassengera Z, Dalcomune D, Turkan H, Vincent JL. Serial determinations of neutrophil CD64 expression for the diagnosis and monitoring of sepsis in critically ill patients. Clin Infect Dis. 2014; 58(6):820-9.
doi: 10.1093/cid/cit936 pmid: 24363321 |
| 38 |
Gibot S, Bene MC, Noel R, Massin F, Guy J, Cravoisy A, et al. Combination biomarkers to diagnose sepsis in the critically ill patient. Am J Respir Crit Care Med. 2012; 186(1):65-71.
doi: 10.1164/rccm.201201-0037OC |
| [1] | Li-wei Duan, Jin-long Qu, Jian Wan, Yong-hua Xu, Yi Shan, Li-xue Wu, Jin-hao Zheng, Wei-wei Jiang, Qi-tong Chen, Yan Zhu, Jian Zhou, Wen-bo Yu, Lei Pei, Xi Song, Wen-fang Li, Zhao-fen Lin. Effects of viral infection and microbial diversity on patients with sepsis: A retrospective study based on metagenomic next-generation sequencing [J]. World Journal of Emergency Medicine, 2021, 12(1): 29-35. |
| [2] | Hai-jiang Zhou, Tian-fei Lan, Shu-bin Guo. Outcome prediction value of National Early Warning Score in septic patients with community-acquired pneumonia in emergency department: A single-center retrospective cohort study [J]. World Journal of Emergency Medicine, 2020, 11(4): 206-215. |
| [3] | Yu-ming Wang, Yan-jun Zheng, Ying Chen, Yun-chuan Huang, Wei-wei Chen, Ran Ji, Li-li Xu, Zhi-tao Yang, Hui-qiu Sheng, Hong-ping Qu, En-qiang Mao, Er-zhen Chen. Effects of fluid balance on prognosis of acute respiratory distress syndrome patients secondary to sepsis [J]. World Journal of Emergency Medicine, 2020, 11(4): 216-222. |
| [4] | Miao Yuan, Ding-yi Yan, Fang-shi Xu, Yi-di Zhao, Yang Zhou, Long-fei Pan. Effects of sepsis on hippocampal volume and memory function [J]. World Journal of Emergency Medicine, 2020, 11(4): 223-230. |
| [5] | Shao-hua Liu, Huo-yan Liang, Hong-yi Li, Xian-fei Ding, Tong-wen Sun, Jing Wang. Effect of low high-density lipoprotein levels on mortality of septic patients: A systematic review and meta-analysis of cohort studies [J]. World Journal of Emergency Medicine, 2020, 11(2): 109-116. |
| [6] | Yi-wen Fan, Shao-wei Jiang, Jia-meng Chen, Hui-qi Wang, Dan Liu, Shu-ming Pan, Cheng-jin Gao. A pulmonary source of infection in patients with sepsis-associated acute kidney injury leads to a worse outcome and poor recovery of kidney function [J]. World Journal of Emergency Medicine, 2020, 11(1): 18-26. |
| [7] | Kimberly A. Chambers, Adam Y. Park, Rosa C. Banuelos, Bryan F. Darger, Bindu H. Akkanti, Annamaria Macaluso, Manoj Thangam, Pratik B. Doshi. Outcomes of severe sepsis and septic shock patients after stratification by initial lactate value [J]. World Journal of Emergency Medicine, 2018, 9(2): 113-117. |
| [8] | Shahin Shadnia, Nasim Zaman, Hossein Hassanian-Moghaddam, Hamed Shafaroodi, Mina Padandar, Mohammad Hasan Rezaeizadeh. Prognostic value of cortisol and thyroid function tests in poisoned patients admitted to toxicology ICU [J]. World Journal of Emergency Medicine, 2018, 9(1): 51-55. |
| [9] | Muhammad Akbar Baig, Hira Shahzad, Erfan Hussain, Asad Mian. Validating a point of care lactate meter in adult patients with sepsis presenting to the emergency department of a tertiary care hospital of a low- to middle-income country [J]. World Journal of Emergency Medicine, 2017, 8(3): 184-189. |
| [10] | Wan-qin Xie, Lin Zhou, Yong Chen, Bin Ni. Circulating microRNAs as potential biomarkers for diagnosis of congenital heart defects [J]. World Journal of Emergency Medicine, 2016, 7(2): 85-89. |
| [11] | Jing Li, Huan Ye, Li Zhao. B-type natriuretic peptide in predicting the severity of community-acquired pneumonia [J]. World Journal of Emergency Medicine, 2015, 6(2): 131-136. |
| [12] | Xue-zhong Xing, Yong Gao, Hai-jun Wang, Shi-ning Qu, Chu-lin Huang, Hao Zhang, Hao Wang, Qing-ling Xiao, Ke-lin Sun. Effect of sedation on short-term and long-term outcomes of critically ill patients with acute respiratory insufficiency [J]. World Journal of Emergency Medicine, 2015, 6(2): 147-152. |
| [13] | Chao Cao, Tao Ma, Yan-fen Chai, Song-tao Shou. The role of regulatory T cells in immune dysfunction during sepsis [J]. World Journal of Emergency Medicine, 2015, 6(1): 5-9. |
| [14] | Wei-ping Sun, Guang-xiong Yuan, Yan-juan Hu, Li-zhen Liao, Lin Fu. Effect of low-dose glucocorticoid on corticosteroid insufficient patients with acute exacerbation of chronic obstructive pulmonary disease [J]. World Journal of Emergency Medicine, 2015, 6(1): 34-39. |
| [15] | Kun Chen, Qiu-xiang Zhou, Hong-wei Shan, Wen-fang Li, Zhao-fen Lin. Prognostic value of CD4+CD25+ Tregs as a valuable biomarker for patients with sepsis in ICU [J]. World Journal of Emergency Medicine, 2015, 6(1): 40-43. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
